Why Clinical Data Warehouse is Essential For Your Clinical Trials?
Data collected in clinical trials are complex, vast and growing uncontrollably with the number of clinical studies, clinical research softwares and vendors present in the clinical research market. For one study, a Clinical research company must work with several different vendors and software including
- CDM/EDC Systems (like Oracle Clinical/RDC, Inform, RAVE, Open Clinica, Clinevo EDC etc.)
- Safety Systems (like Argus, ArisG, Clinevo Safety, etc.)
- CTMS Systems (like Siebel Clinical, Clintrace, Clinevo CTMS)
- SAS-Based Systems
- Cloud-Based Systems
- Legacy Systems
- Spreadsheets
- Local and Central Lab Systems
- EMR/EHR Systems
- Regulatory Submission Systems.
Can you store, access and interact with all your clinical trial data in one place?
Data from all these different systems/sources being in different formats, makes impossible to get all the data in one place.!
What is a Clinical Data Warehouse?
A Clinical Data Warehouse or a Clinical Data Repository is a real-time database that consolidates data from a variety of clinical, safety and non-clinical sources to present a unified view.
All the data like Clinical Data, Operations Data, Safety Data, Lab Data and other data of one study can be available in one place for review, reporting and analytics. A Business intelligence tool in combination with a Data warehouse can enable you to know the progress of the trial in real time and enables you to monitor and take right decisions at the right time.
- Storage
- Review
- Standardization
- Aggregation
- Analysis
- Submission
Data warehousing enables you to perform
- Cross study analysis
- Data harmonization
- Data reconciliation
- Data recording
- SDTM transformation
- Data Aggregation and
- any business processes
Purpose of a Clinical Data Warehouse:
Purpose of Clinical Data Warehouse lies somewhere in its definition itself i.e. a database created by combining data that is gathered through various sources that can be of different types and formats (e.g. oracle, SAS, text, sql, xml etc.).
In a nutshell, Data Warehouse is a process of collecting data, transforming it, loading into single database and then using a BI (Business Intelligence) tool to answer your analytical queries and prediction of any further questions that may arise are helpful to your domain or business.
Benefits of a Clinical Data Warehouse:
Below are a few benefits of implementing a Clinical Data Warehouse for your Clinical Trials:
- Improving Visibility of Data / All-in-One Platform:Clinical research organizations process data in different systems, which support the various business processes. In order to create an overall picture of clinical, operations, safety, non-clinical, lab and other data for sponsors and CROs – thus creating a single version of the truth – the data must come together in one place and made compatible. Both external (from the environment) and internal data (from various systems) should merge into the data warehouse and then be grouped. Therefore, having a single source to answers all your queries.
- Improved Performance: One could use an already existing operational database if there is only single database for all the data, yet there few constraints like performance which degrade for both operational processes and reporting processes. Therefore, we create a database tuned and optimized database which will be ready to answer queries which require to bring huge amount of data and analysis.
- Increase Data Quality: Users frequently overestimate the quality of data in the source systems. Unfortunately, source systems often contain data of poor quality. When we use a data warehouse, we can greatly improve the data quality, either through – were possible – correcting the data whilst loading or by tackling the problem at its source.
- Faster Decision Making and More advanced Reporting: The structure of both data warehouses enables end users to report in a flexible manner and to quickly perform interactive analysis based on various predefined angles. They may, for example, with a single mouse click jump from year level – to quarter – to month level and quickly switch between the clinical data and the operational data whereby the indicator remains fixed. In this way, end users can juggle with the data and thus quickly gain knowledge about business operations and KPIs (Key Performance Indicator). Some metrics like – Which Site is not performing well? Which Investigator is not performing? Which patient is not regular for trials? Can be zeroed in in no time to find out the bottle necks of a trial and take necessary action time to avoid delays in the trails because of non-performing factors.
- Regulatory Compliance: Clinical data warehouse stores audits of every action and stores data snapshot of data of different timestamps and provides version controls for the loads and programs which can help convincing the auditors on the data security, data handling and single source of truth.
About Clinevo Data Warehouse:
Clinevo Data Warehouse is an easy to use, secured, regulatory compliance clinical trials data repository & warehouse to acquire, store, transform, consolidate and report diverse data of clinical trials in one place.
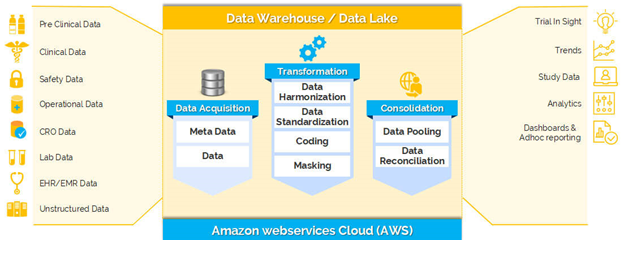
Features of Clinevo Data Warehouse:
- One Clinical Data Repository:
- Holistic view of all clinical, safety and operational data of a clinical trial in one place
- Wide Range of Tech Type Integrations:
- Oracle, SAS, Text/CSV, XML, SQL Server, web services Imports
- Oracle PL/SQL and SAS programs
- SAS, TEXT, XML, Oracle Extracts
- Inbuilt Business Process
- Data Harmonization – Harmonize data from different source structure to one target structure
- Data pooling – To the database schema (or) to remote DB with security
- Data masking – supports various approaches
- Increased Efficiency and Automation
- Robust incremental refresh to get the data refreshed within 4 hours from a heavy Source system
- Incremental data refresh for non-primary key tables
- Incremental data refresh based on data element group
- Parallel Data Loading
- Configurable Automated Data Loads
- Clinical Metadata Repository (MDR) Integration
- Any Clinical MDR systems (COTS) or Legacy systems can be integrated with CLIDAC directly (or) thru web services to get the metadata.
- Audit/ Regulatory Compliant
- 21CFR part 11 regulatory compliance
- Audit trail for every operation
- Complete user tracking
Read more about the features of Clinevo Data Warehouse:



